RBSE Solutions for Class 8 Science Chapter 4 Materials: Metals and Non-Metals
Rajasthan Board RBSE Solutions for Class 8 Science Chapter 4 Materials: Metals and Non-Metals Textbook Exercise Questions and Answers.
Rajasthan Board RBSE Solutions for Class 8 Science in Hindi Medium & English Medium are part of RBSE Solutions for Class 8. Students can also read RBSE Class 8 Science Important Questions for exam preparation. Students can also go through RBSE Class 8 Science Notes to understand and remember the concepts easily. Browsing through class 8 science chapter 14 extra questions that includes all questions presented in the textbook.
RBSE Class 8 Science Solutions Chapter 4 Materials: Metals and Non-Metals
RBSE Class 8 Science Materials: Metals and Non-Metals InText Questions and Answers
Page 47
Question 1.
Does copper also get rusted? I have seen a greenish deposit on the surface of copper vessels.
Or
What happens when a copper vessel is exposed to moist air for a long time.
Or
Due to which, chemical substance the green coloured layer is deposited on the upper region of copper jug? Explain with chemical equation for this formation.
Answer:
When a copper vessel or copper jug is exposed to moist air for long time it acquires a dull green coating. The green material is a mixture of copper hydroxide (Cu(OH)2) and copper carbonate (CuCO3). The reaction or chemical equation for this is as follows:
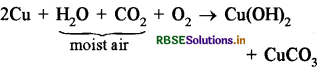
Copper + Water + Carbondioxide + Oxygen → Copper Hydroxide + Copper Carbonate

Page 52
Question 2.
I heard that magnesium is found in plants. In what form is it found in them?
Answer:
Magnesium is found in green plants in the form of chlorophyll.
Question 3.
The doctor reported iron deficiency in my body.Where is iron in my body?
Answer:
Iron is an element which is present in every cell of our body. In the human body mainly iron is present in red blood corpuscles.
RBSE Class 8 Science Materials: Metals and Non-Metals Textbook Questions and Answers
Question 1.
Which of the following can be beaten into thin sheets?
(a) Zinc
(b) Phosphorus
(c) Sulphur
(d) Oxygen
Answer:
(a) Zinc

Question 2.
Which of the following statements is correct?
(a) All metals are ductile.
(b) All non - metals are ductile.
(c) Generally, metals are ductile.
(d) Some non - metals are ductile.
Answer:
(c) Generally, metals are ductile.
Question 3.
Fill in the blanks:
(a) Phosphorus is a very ................. non - metal.
(b) Metals are ................. conductors of heat and .................
(c) Iron is ................. reactive than copper.
(d) Metals react with acids to produce ................. gas.
Answer:
(a) active
(b) good, electricity
(c) more
(d) hydrogen.
Question 4.
Mark 'T' if the statement is true and 'F' if it is false.
(a) Generally, non - metals react with acids ( )
(b) Sodium is a very reactive metal ( )
(c) Copper displaces zinc from zinc sulphate solution ( )
(d) Coal can be drawn into wires ( )
Answer:
(a) F
(b) T
(c) F
(d) F

Question 5.
Some properties are listed in the following Table. Distinguish between metals and non - metals on the basis of these properties.
|
Properties |
Metals |
Non-metals |
|
1. Appearance |
|
|
|
2. Hardness |
|
|
|
3. Malleability |
|
|
|
4. Ductility |
|
|
|
5. Heat Conduction |
|
|
|
6. Conduction of Electricity |
|
|
Answer:
|
Properties |
Metals |
Non-metals |
|
1. Appearance |
Lustrous |
Not Lustrous |
|
2. Hardness |
Hard |
Not Hard |
|
3. Malleability |
Malleable |
Not Malleable |
|
4. Ductility |
Ductile |
Not Ductile |
|
5. Heat Conduction |
Good Conductor of heat |
Bad Conductor of heat |
|
6. Conduction of Electricity |
Good Conductor of Electricity |
Bad Conductor of Electricity |
Question 6.
Give reasons for the following:
(a) Aluminium foils are used to wrap food items.
(b) Immersion rods for heating liquids are made up of metallic substances.
(c) Copper cannot displace zinc from its salt solution.
(d) Sodium and potassium are stored in kerosene.
Answer:
(a) Aluminium foils are used to wrap food items because aluminium is malleable. It means it can be beaten into thin sheets and foils. It does not react with food items also.
(b) Immersion roads for heating liquids are made up of metallic substances because metals are good conductor of heat and electricity.
(c) Copper cannot displace zinc from its salt solution because copper is less reactive than zinc.
(d) Sodium and potassium are very reactive. They react vigorously with oxygen and water and a lot of heat is generated in the reaction. They are therefore, stored in kerosene.

Question 7.
Can you store lemon pickle in an aluminium utensil? Explain.
Answer:
No, lemon pickle is acidic in nature and acidic substance can not be stored in an aluminium utensil because it reacts with acid and forms poisonous substances which makes food poisonous.
Question 8.
Match the substances given in Column A with their uses given in Column B.
|
Column A |
Column B |
|
(i) Gold |
(a) Thermometers |
|
(ii) Iron |
(b) Electric wire |
|
(iii) Aluminium |
(c) Wrapping food |
|
(iv) Carbon |
(d) Jewellery |
|
(v) Copper |
(e) Machinery |
|
(vi) Mercury |
(f) Fuel |
Answer:
|
Column A |
Column B |
|
(i) Gold |
(d) Jewellery |
|
(ii) Iron |
(e) Machinery |
|
(iii) Aluminium |
(c) Wrapping food |
|
(iv) Carbon |
(f) Fuel |
|
(v) Copper |
(b) Electric wire |
|
(vi) Mercury |
(a) Thermometers |
Question 9.
What happens when:
(a) Dilute sulphuric acid is poured on a copper plate?
(b) Iron nails are placed in copper sulphate solution?
Write word equations of the reactions involved.
Answer:
(a) When dilute sulphuric acid is poured on a copper plate, copper sulphate and hydrogen gases are produced.
Copper + Sulphuric acid → Copper sulphate + Hydrogen
(b) When iron nails are placed in copper sulphate solution, copper is displaced from copper sulphate solution. Iron sulphate and copper are obtained in this reaction.
Copper sulphate + Iron (Nails) → Iron sulphate + Copper

Question 10.
Saloni took a piece of burning charcoal and collected the gas evolved in a test tube.
(a) How will she find the nature of the gas?
(b) Write down word equations of all the reactions taking place in this process.
Answer:
(a) The gas evolved from burning of charcoal is carbon dioxide which forms carbonic acid on dissolution in water. Blue litmus becomes red when dipped in this which proves the acidic nature of the solution.
(b) When charcoal is binned it reacts with oxygen which gives carbon dioxide, which is acidic.
Charcoal + Oxygen → Carbon dioxide
Question 11.
One day Reeta went to a jeweller's shop with her mother. Her mother gave an old gold jewellery to the goldsmith to polish. Next day when they brought the jewellery back, they found that there was a slight loss in its weight. Can you suggest a reason for the loss in weight?
Answer:
Goldsmith use the aquaregia solution for the cleaning of jewellery. Gold dissolves in this solution so the weight of Reeta's mother's jewellery becomes less. Aquaregia is prepared by mixing hydrochloric acid (3 part) and nitric acid (1 part).

- RBSE Class 8 Science Notes in Hindi & English Medium Pdf Download
- RBSE Class 8 Science Important Questions in Hindi Medium & English Medium
- RBSE Solutions for Class 8 Science in Hindi Medium & English Medium
- RBSE Class 8 Science Important Questions Chapter 14 Chemical Effects of Electric Current
- RBSE Class 8 Science Notes Chapter 16 Light
- RBSE Class 8 Science Notes Chapter 1 Crop Production and Management
- RBSE Class 8 Science Notes Chapter 2 Microorganisms: Friend and Foe
- RBSE Class 8 Science Notes Chapter 3 Synthetic Fibres and Plastics
- RBSE Class 8 Science Notes Chapter 4 Materials: Metals and Non-Metals
- RBSE Class 8 Science Notes Chapter 5 Coal and Petroleum
- RBSE Class 8 Science Notes Chapter 6 Combustion and Flame