RBSE Solutions for Class 11 Physics Chapter 13 Kinetic Theory
Rajasthan Board RBSE Solutions for Class 11 Physics Chapter 13 Kinetic Theory Textbook Exercise Questions and Answers.
Rajasthan Board RBSE Solutions for Class 11 Physics in Hindi Medium & English Medium are part of RBSE Solutions for Class 11. Students can also read RBSE Class 11 Physics Important Questions for exam preparation. Students can also go through RBSE Class 11 Physics Notes to understand and remember the concepts easily.
RBSE Class 11 Physics Solutions Chapter 13 Kinetic Theory
RBSE Class 11 Physics Kinetic Theory Textbook Questions and Answers
Question 13.1.
Estimate the fraction of molecular volume to the actual volume occupied by oxygen gas at STP. Take thte diameter of an oxygen molecule to be 3 Å.
Solution:
Given: Diameter, d = 3 Å
∴ r = \(\frac{d}{2}=\frac{3}{2}\) Å = \(\frac{3}{2}\) x 10-8 cm
Molecular volume, V = \(\frac{4}{3}\)πr3 NA
= \(\frac{4}{3}\) x \(\frac{22}{7}\) x (1.5 x 10-8)3 x 6.023 x 1023
= 8.25 cm3
Actual volume occupied by one mole of oxygen at STP,
V' = 22400 cm3
∴ \(\frac{V}{V^{\prime}}=\frac{8.52}{22400}\) = 3.8 x 10-4

Question 13.2.
Molar volume is the volume occupied by 1 mole of any (ideal) gas at standard temperature and pressure (STP: 1 atmospheric pressure, 0°C). Show that it is 22.4 litres.
Solution:
Ideal gas equation (for n = 1)
PV = RT
∴ V = \(\frac{R T}{P}\)
Here, R = 8.31 J mol-1 K-1
T = 273 K
P = 1 atm = 1.013 x 105 N-m-2
∴ V = \(\frac{8.31 \times 273}{1.013 \times 10^5}\) = 0.0224 m3
= 22.4 L
Question 13.3.
Figure shows plot of \(\frac{P V}{T}\) versus P for 1.00 x 10-3 kg of oxygen gas at two different temperature.
(a) What does the dotted plot signify?
(b) Which is true: T1 > T2 or T1 < T2?
(c) What is the value of \(\frac{P V}{T}\) where the curve meet on the y-axis?
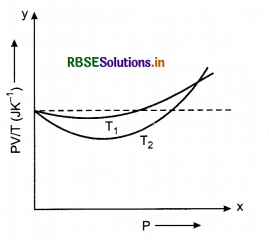
(d) If we obtain similar plots for 1.00 x 103 kg of hydrogen, would we get the same value of \(\frac{P V}{T}\) at the point where the curves meet on the y-axis? If not, what mass of hydrogen yields the same value of \(\frac{P V}{T}\) (for low pressure high temperature region of the plot)? (Molecular mass of H2 = 2.02 u, of O2 = 32.0 u, R = 8.31 J mol-1 K-1.)
Solution:
(a) The dotted plot signifies the ideal gas behaviour.
(b) The curve at temperature T1 is closer to the dotted plot than that for T2.
∴ T1 > T2
(c) When the two curves meet on the y-axis, the value of \(\frac{P V}{T}\) = nR
= \(\frac{1}{32}\) x 8.31 JK-1
= 0.26 JK-1
(d) If we obtained similar plots for 1.00 x 10-3 kg of hydrogen, then we would not get the same value of \(\frac{P V}{T}\) at the point where the curves meet on the y-axis. It is so because the molar mass of hydrogen is different from that of oxygen.
The mass of hydrogen can be yielded for the same value of \(\frac{P V}{T}\) from

Question 13.4.
An oxygen cylinder of volume 30 litres has an initial gauge pressure of 15 atm and a temperature of 27°C. After some oxygen is withdrawn from the cylinder, the gauge pressure drops to 11 atm and its temperature drops to 17°C. Estimate'the mass of oxygen taken out of the cylinder (R = 8.31 J mol-1 K-1, molecular mass of O2 = 32 u).
Solution:
Initially,
in the oxygen cylinder,
V1 = 30 L = 30x 10-3 m3
P1 = 15 atm = 15 x 1.01 x 105 Pa
T1 = (27 + 273) K = 300 K
For n1 moles,
P1V1 = n1RT1
⇒ n1 = \(\frac{P_1 V_1}{R T_1}=\frac{15 \times 1.01 \times 10^5 \times 30 \times 10^{-3}}{8.3 \times 300}\)
= 18.253
Molar mass of ooxygen gas, M = 32 g
Mass of oxygen in the cylinder, m1 = n1M
= 18.253 x 32 = 584.18
∴ Mass of oxygen, m2 = 13.847 x 32 = 435.1 g
∴ Mass of oxygen gas withdrawn
= m1 - m2 = (584.1 - 453.1)g = 131 g

Question 13.5.
An air bubble of volume 1.0 cm3 rises from the bottom of a lake 40 m deep at a temperature of 12°C. To what volume does it grow when it reaches the surface, which is at a temperature of 35°C? Given 1 atm = 1.01 x 105 Pa.
Solution:
V1 = 1 cm3 = 1 x 10-6 m3
T1 = 12°C = (12 + 273) = 285 K
P1 = 1 atm + h1ρg
= 1.01 x 105 + 40 x 103 x 9.8
= 493000 Pa
When air bubble reaches the surface,
V2 = ?
T2 = 35° C = (35 + 273) K = 308 K
P2 = 1 atm = 1.01 x 105 Pa

Question 13.6.
Estimate the total number of air molecules (inclusive of oxygen, nitrogen, water vapour and other constituents) in a room of capacity 25.0 m3 at a temperature of 27°C and 1 atm pressure. (Boltzmann constant = 1.38 x 10-23 JK-1)
Solution:
V = 25.0 m3,
T = 27° C = (27 + 273) K = 300 K
Boltzmann constant, kB = 1.38 x 10-23 JK-1
PV = nRT
= n x (NkB) x T
= nN x kB x T
= N'kBT (here nN = N')
N' is total number of air molecules
∴ N' = \(\frac{P V}{k_B T}=\frac{1.01 \times 10^5 \times 25}{1.38 \times 10^{-23} \times 300}\) = 6.10 x 1026
Question 13.7.
Estimate the average thermal energy of a helium atom at (i) room temperature (27°C), (ii) the temperature on the surface of the Sun (6000 K), (iii) the temperature of 10 million Kelvin (the typical core temperature in the case of a star).
Solution:
(i) Here, T = 27° = (27 + 273) K = 300 K
Average thermal energy,
E = \(\frac{3}{2}\)kT = \(\frac{3}{2}\) x 1.38 x 10-23 x 300
= 6.2 x 10-21 J
(ii) At T = 6000 K
Average thermal energy,
E = \(\frac{3}{2}\)kT = \(\frac{3}{2}\) x 1.38 x 10-23 x 6000
= 1.24 x 10-19 J
(iii) At T = 107 K
Average thermal energy, E = \(\frac{3}{2}\)kT
= \(\frac{3}{2}\) x 1.38 x 10-23 x 107
= 2.1 x 10-16 J
Question 13.8.
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monoatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic). Do the vessels contain equal number of respective molecules? Is the root mean square speed of molecules the same in the three cases? If not, in which case is vrms the largest?
Solution:
As the three vessels at the same temperature and pressure have equal capacity (volume). So, according to the Avogadro’s law, the three vessels will have equal number of molecules, equal to NA = 6.023 x 1023.
∴ vrms = \(\sqrt{\frac{3 k T}{m}}\)
∴ RMS speed of molecules will not be the same in the three cases.
vrms will be largest in case of neon, as it has the smallest mass.
Question 13.9.
At what temperature is the root mean square speed of an atom in an argon gas cylinder equal to the r.m.s. speed of a helium gas atom at -20°C? (Atomic mass of Ar = 39.9 u, of He = 4.0 u)
Solution:
Given: Atomic mass of Ar, M = 39.9 u
Atomic mass of He, M' = 4.0 u
Let the temperatures of Argon and Helium gas atoms be T and T'.
T' = (-20 + 273) K = 253 K

Question 13.10.
Estimate the mean free path and collision frequency of a nitrogen molecule in a cylinder containing nitrogen at 2 atm and temperature 17°C. Take the radius of a nitrogen molecule to be roughly 1.0 A. Compare the collision time with the time the molecule moves freely between two successive collisions. (Molecular mass of N2 = 28.0 u).
Solution:
P = 2 atm = 2 x 1.013 x 105 Nm-2
= 2.026 x 105 Nm-2
T = 17° C = (17 + 273) K = 290K
d = 2 x 1 Å = 2 x 10-10 m
kB = 1.38 x 10-23 J mol-1 K-1
M = 28 x 10-3 kg

Collision frequency = Number of collisions per second
= \(\frac{v_{\mathrm{rms}}}{\lambda}=\frac{508.24}{1.11 \times 10^{-7}}\) = 4.58 x 109 s-1
Additional Exercise
Question 13.11.
A meter long narrow bore held horizontally (and closed at one end) contains a 76 cm long mercury thread which traps a 15 cm column of air. What happens if the tube is held vertically with the open end at the bottom?
Solution:
When the meter long narrow bore is held horizontally, the mercury thread of length 76 cm. traps air (in length) = 15 m. A length of 9 cm of the bore will be left at the open end. Let the area of the cross-section of the bore be 1 cm2.
P1 = 76 cm and = 15 cm2
When the bore is held vertically, 15 cm air gets another 9 cm air. Now, suppose h cm of mercury flow out to balance the atm pressure.
The heights of air column and mercury column will be (24 + h) cm and (76 - h) cm respectively.
The pressure of air = 76 - (76 - h) = h cm of Hg
V2 = (24 + h) cm3
P2 = h cm
If temperature is constant, then
P1V1 = P2V2
⇒ 76 x 15 = h x (24 + h)
⇒ 1140 = 24h + h2
⇒ h2 + 24h - 1140 = 0
⇒ h = \(\frac{-24 \pm \sqrt{24^2+4 \times 1140}}{2}\)
∴ h = 23.8 cm or -47.8 cm
h cannot be negative. So, h = 23.8 cm
Thus, of the tube os held vertically with the open end at bottom, then 23.8 cm of Hg flows out.
Question 13.12.
From a certain apparatus, the diffusion rate of hydrogen has an average value of 28.7 cm3 s-1. The diffusion of another gas under the same conditions is measured to have an average rate of 7.2 cm3 s-1. Identify the gas.
[Hints: Use Graham's Law of diffusion \(\frac{R_1}{R_2}\) = (\(\frac{M_1}{M_2}\))1/2, where R1, R2 are diffusion rates of gases 1 and 2 and M1 and M2 their respective molecular masses. The law is a simple consequence of kinetic theory.]
Solution:
According to Graham's law of diffusion
\(\frac{r_1}{r_2}=\sqrt{\frac{M_2}{M_1}}\)
Here, Diffusion rate of hydrogen,
r1 = 28.7 cm3 s-1
Diffusion rate of unknown gas,
r2 = 7.2 cm3 s-1
Molecular mass of hydrogen, M1 = 2 u
∴ \(\frac{28.7}{7.2}=\sqrt{\frac{M_2}{2}}\)
⇒ M2 = \(\left(\frac{28.7}{7.2}\right)^2 \times 2\) = 32
The unknown gas is oxygen, as its molar mass is 32 u.

Question 13.13.
A gas in equilibrium has uniform density and pressure throughout its volume. This is strictly true only if there are no external influences. A gas column under gravity, for example, does not have uniform density (and pressure). As you might expect, its density decreases with height. The precise dependence is given by the so called Taw of atmospheres’:
n2 = n1 exp.[\(\frac{-m g\left(h_2-h_1\right)}{k_B T}\)]
where n1, n2 refer to number density at heights h1 and h2 respectively. Use this relation to derive the equation for sedimentation equilibrium of suspension in a liqiud column:
n2 = n1 exp.[-mg NA (ρ - ρ') \(\frac{\left(h_2-h_1\right)}{\rho R T}\)]
where ρ is the density of the suspended particle, and ρ' that of surrounding medium.
[NA is Avogadro's number, and R is the universal gas constant.]
Solution:
n2 and n1 are the number densities of particles at heights h2 and h1 respectively.
Let V, ρ and m be the average volume, density and mass respectively of the suspended particle. Let ρ' and m' be the density and mass of the liquid respectively.
According to Archimedes' principle,
Effective weight of suspended particle = Actual weight - Weight of liquid displaced

Question 13.14.
Given below are densities of some solids and liquids. Give rough estimates of the size of their atoms:
|
Substance |
Atomic Mass (u) |
Density (103 kg m-3) |
|
Carbon (diamond) |
12.01 |
2.22 |
|
Gold |
197.00 |
19.32 |
|
Nitrogen (liquid) |
14.01 |
1.00 |
|
Lithium |
6.94 |
0.53 |
|
Fluorine (liquid) |
19.00 |
1.14 |
[Hint: Assume the atoms to be 'tightly packed' in a solid or liquid phase, and use the known value of Avogadro's number. You should, however, not take the actual numbers you obtain for various atomic sizes too literally. Because of the crudeness of the tight packing approximation, the results only indicate that atomic sizes are in the range of a few Å]
Solution:
Let r be the radius of the atom, then,
Volume of each atom = \(\frac{4}{3}\)πr3
Volume of all atoms in one mole of substance
= \(\frac{4}{3}\)πr3 x N = \(\frac{M}{\rho}\)
∴ r = \(\left[\frac{3 M}{4 \pi \rho N}\right]^{1 / 3}\)
For carbon (C)
M = 12.01 x 10-3 kg,
ρ = 2.22 x 103 kg m-3
∴ r = \(\frac{3 \times 12.01 \times 10^{-3}}{4 \times 3.14 \times 2.22 \times 10^3 \times 6.023 \times 10^{23}}\)
= 1.29 Å
Similarly, for Gold, rG = 1.59 Å
for Nitrogen (liquid), rN = 1.77 Å
for Lithium, rL = 1.73 Å
and, for Fluorine (liquid), rF = 1.88 Å

- RBSE Class 11 Physics Important Questions Chapter 4 Motion in a Plane
- RBSE Solutions for Class 11 Physics Chapter 15 Waves
- RBSE Solutions for Class 11 Physics Chapter 14 Oscillations
- RBSE Solutions for Class 11 Physics Chapter 12 Thermodynamics
- RBSE Solutions for Class 11 Physics Chapter 11 Thermal Properties of Matter
- RBSE Solutions for Class 11 Physics Chapter 10 Mechanical Properties of Fluids
- RBSE Solutions for Class 11 Physics Chapter 9 Mechanical Properties of Solids
- RBSE Solutions for Class 11 Physics Chapter 8 Gravitation
- RBSE Solutions for Class 11 Physics Chapter 7 System of Particles and Rotational Motion
- RBSE Solutions for Class 11 Physics Chapter 6 Work, Energy and Power
- RBSE Solutions for Class 11 Physics Chapter 5 Laws of Motion